Phase I/II Clinical Trials Investigating PS cell therapy for Parkinson's disease.
Background:
Parkinson’s disease is caused by a loss of dopamine neurons, causing motor symptoms. Initial cell therapies using fetal tissues showed promise but had complications and ethical concerns.
1. Pluripotent stem (PS) cells emerge as a promising alternative for developing safe and effective treatments in 2025
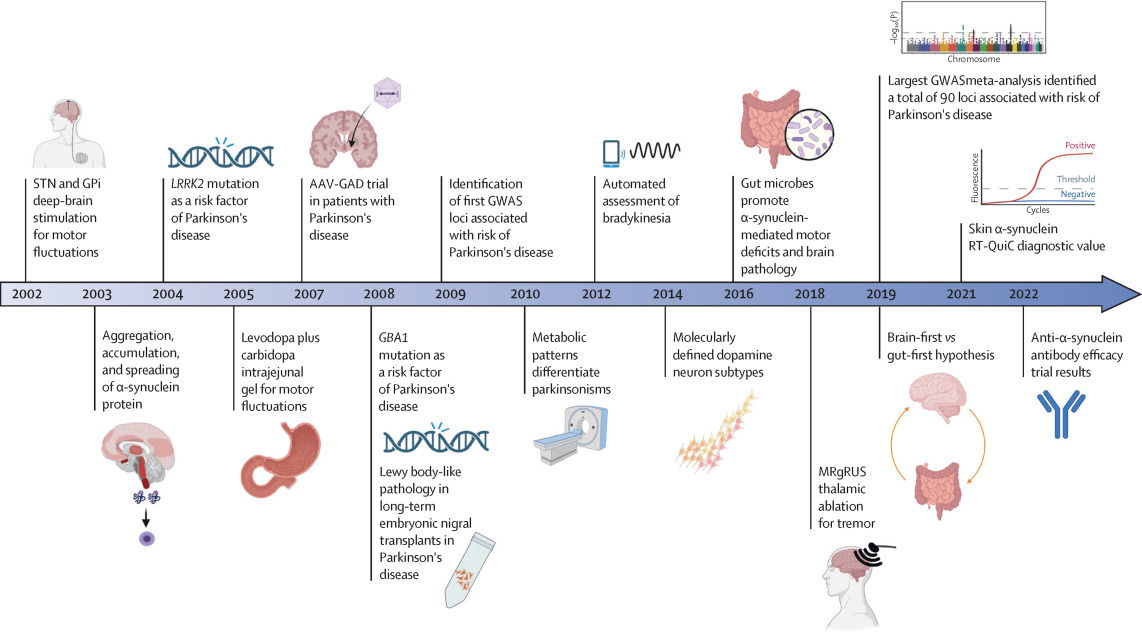
Image credit: Major advances in Parkinson's disease over the past two decades and future research directions
Obeso, Jose A et al.The Lancet Neurology, Volume 21, Issue 12, 1076 - 1079
2025 Update:
In a phase I/II trial at Kyoto University Hospital, seven patients (ages 50–69) received bilateral transplantation of dopaminergic progenitors derived from induced PS (iPS) cells. Primary outcomes focused on safety and adverse events, while secondary outcomes assessed motor symptom changes and dopamine production for 24 months. There were no serious adverse events, with 73 mild to moderate events.
Among six patients subjected to efficacy evaluation, four showed improvements in the Movement Disorder Society Unified Parkinson’s Disease Rating Scale (MSD-UPDRS) part III OFF score, and five showed improvements in the ON scores. This scale is often used in clinical settings and in research.
The average changes of all six patients were 9.5 (20.4%) and 4.3 points (35.7%) for the OFF and ON scores, respectively. Hoehn–Yahr stages improved in four patients. Fluorine-18-l-dihydroxyphenylalanine (18F-DOPA) influx rate constant (Ki) values in the putamen increased by 44.7%, with higher increases in the high-dose group. Other measures showed minimal changes. This trial (jRCT2090220384) demonstrated that allogeneic iPS-cell-derived dopaminergic progenitors survived, produced dopamine and did not form tumours, therefore suggesting safety and potential clinical benefits for Parkinson’s disease.
See updated paper: Sawamoto, N., Doi, D., Nakanishi, E. et al. Phase I/II trial of iPS-cell-derived dopaminergic cells for Parkinson’s disease. Nature (2025). https://doi.org/10.1038/s41586-025-08700-0